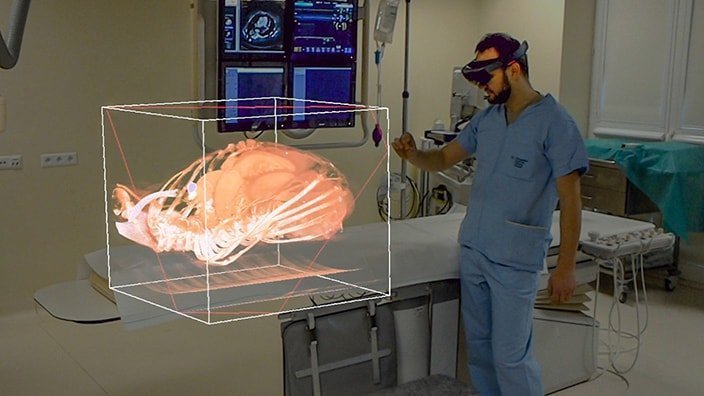
NewConnect-listed Polish company MedApp is stepping up its activities in the world’s most lucrative medical market. Following a series of business meetings in the US, CEO David Odrakiewicz is signalling a real chance of a commercial breakthrough. The key to success is expected to be CarnaLife Holo, a proprietary solution using augmented reality (AR) for 3D visualisation of medical data, which already has the necessary FDA certification.
MedApp’s US offensive is based on building relationships with key players in the health-tech ecosystem. The company’s representatives have actively participated in prestigious industry events such as Sages and BioFlorida, where they presented the technology to surgeons, innovators and medical executives. Of particular note is the visit to AdventHealth – one of the largest independent medical networks in the US. For the Polish company, this institution’s openness to modern infrastructure and innovation is a litmus test of the needs of a market that is increasingly shifting towards digital medicine and precision procedure planning.
MedApp’s strategy, however, goes beyond just selling software. The company is currently in talks with a distribution partner to help scale operations on the continent. A key figure in this process is Habeel Gazi, COO of MedApp USA, who is tasked with building local structures and ‘opening doors’ that have so far remained closed to Central European players.
An interesting business thread is a potential collaboration with Snke, a company working on AR goggles dedicated to the medical sector. This hardware-software synergy could significantly raise the barrier to entry for competitors and offer doctors a complete tool for real-time data visualisation.
MedApp does not treat the US merely as an export destination, but as the focal point of its long-term strategy. Having FDA certification removes the biggest regulatory barrier, and being present in state-of-the-art centres such as AdventHealth confirms that Polish 3D technology is in line with US medical standards. Success in this market will depend on the speed of converting established contacts into hard contracts, which in the complex US healthcare system is an arduous process, but – as the management’s declarations show – currently a priority.